Clinical Architecture
Making sense of what’s next.
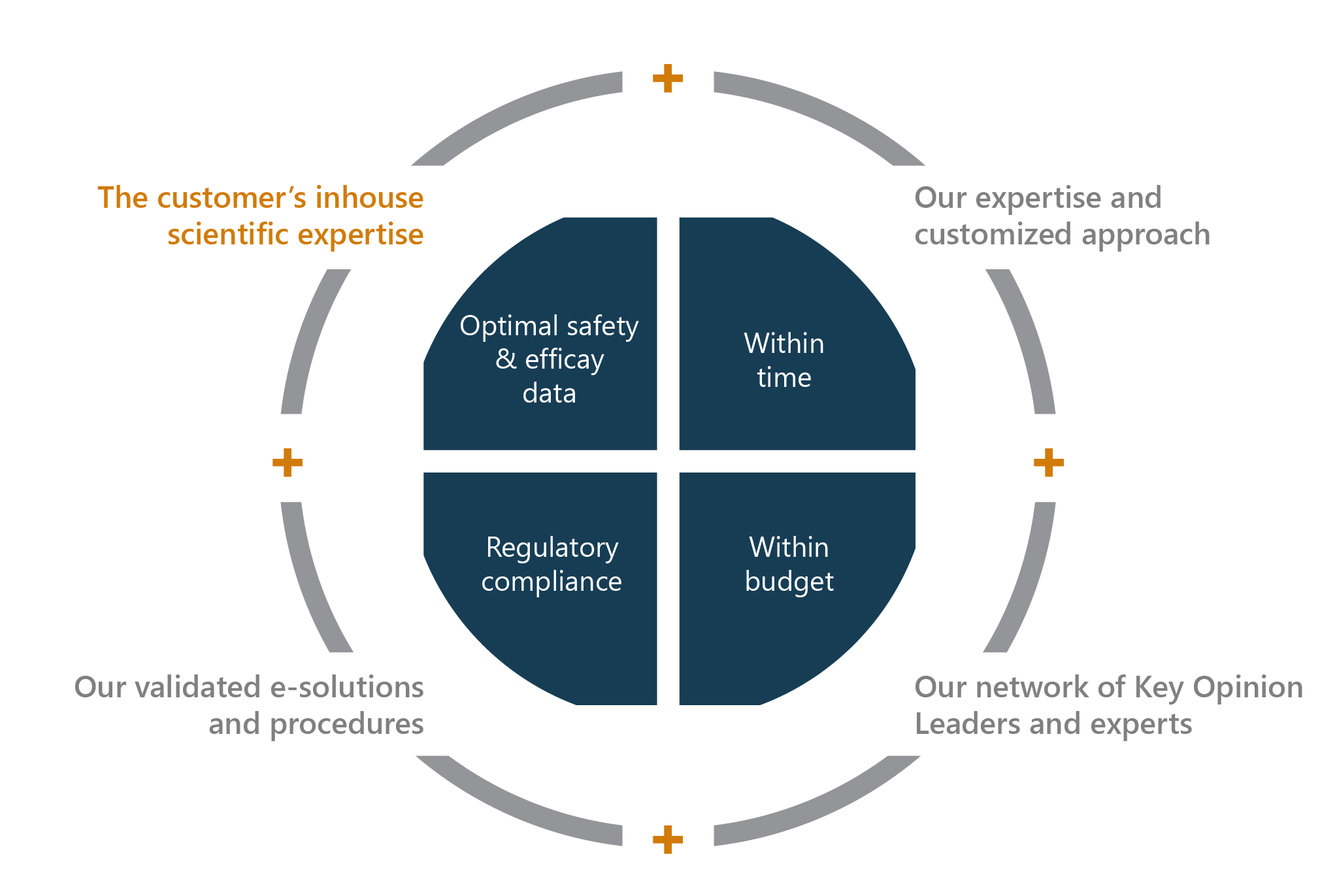
Ambitious projects require an excellent planning. At Sillar Clinical, we guide you in your clinical development strategy from a to z.
Our multitalented team is ready to design and supervise your plan until a successful implementation. We provide you all the necessary building blocks and experts that suit your needs. Our strategists, CMC specialists, regulatory profiles,… are at your disposal to complement your in-house expertise. From Proof of Concept (POC) to Phase IV studies, we make your innovation tick.
Our Clinical Architecture services
We think with you and design your clinical development plan. We provide you all the necessary building blocks that suit all your clinical development phases. Our multitalented team is ready to build and supervise your plan until a successful implementation.
-
Full project management
Each project has his own dedicated Project Manager. Managing all internal and external stakeholders until a successful realization of your project.
-
Clinical development and regulatory strategy
Our dedicated experts and consultants provide you with high-level strategic clinical development services, from clinical trial design, protocol development to full regulatory strategy and submission.
-
Key Opinion Leader network
We help you identifying the right KOL's for your project throughout out strong network.
-
Vendor selection and management
Our experts assist you in selecting the best vendors needed to successfully implement your clinical development plans. Whether it be manufacturing, distribution, non-clinical CRO's, Biostatistics, ... we manage them from the contract phase until the end of your project.
-
Cross-functional expert team
Sillar Clinical has developed an expert team covering multiple needed disciplines, ranging from CMC specialists, Regulatory experts, Safety experts, Biostatisticians over Medical Writers to Clinical Development specialist. They become part of your team, working as one and dedicated to guarantee success.
